Active Ingredients: How Generics Match Brand-Name Drugs in Effectiveness and Cost
 Feb, 24 2026
Feb, 24 2026
When you pick up a prescription, you might see two options: a brand-name pill with a familiar logo, or a cheaper generic with a different color and shape. Many people wonder - are they really the same? The short answer is yes. Active ingredients in generic drugs are identical to those in brand-name drugs. The science, the regulation, and real-world outcomes all point to one thing: generics work just as well.
What Exactly Is an Active Ingredient?
The active ingredient is the part of a medicine that actually does the work. For example, in the blood pressure pill Norvasc, the active ingredient is amlodipine. In the antidepressant Zoloft, it’s sertraline. These chemicals interact with your body to produce the desired effect - lowering blood pressure, reducing anxiety, controlling blood sugar, and so on. Generic drugs contain the exact same active ingredient, in the same amount, and delivered the same way - whether it’s a pill, injection, or inhaler. The FDA requires that generics match the brand-name drug in strength, dosage form, and how it’s taken. That means if your doctor prescribes 10 mg of amlodipine, the generic you get will also deliver 10 mg of amlodipine. No more, no less.How Do We Know Generics Work the Same?
It’s not just a claim. The FDA demands proof. Before a generic drug hits the market, manufacturers must run bioequivalence studies. These tests measure how quickly and how much of the drug enters your bloodstream. The generic’s maximum concentration (Cmax) and total exposure (AUC) must fall within 80% to 125% of the brand-name drug’s numbers. That’s not a wide margin - it’s tight enough to ensure real clinical equivalence. This isn’t theoretical. A landmark 2019 study in JAMA Internal Medicine looked at over 3.5 million patients using generics for diabetes, high blood pressure, osteoporosis, and depression. The results? No difference in outcomes. People taking generic metformin had the same HbA1c levels as those on Glucophage. Those on generic amlodipine lowered their blood pressure just as well as those on Norvasc. Even in psychiatric care, generic escitalopram performed identically to Lexapro. The FDA’s Orange Book lists every approved drug and its therapeutic equivalence rating. If a generic has an “A” rating, it means it’s interchangeable with the brand. Over 90% of all generic drugs in the U.S. carry this rating.Why Are Generics So Much Cheaper?
The price difference isn’t because generics are inferior. It’s because they don’t have to repeat expensive clinical trials. When a brand-name drug is first developed, the company spends years and hundreds of millions of dollars on research, testing, and FDA approval. Once the patent expires - usually after 20 years - other companies can make copies. They don’t need to prove the drug works again. They only need to prove their version behaves the same way in the body. That cuts costs dramatically. According to Brown University Health, generics cost on average 85% less than brand-name versions. Metformin (generic for Glucophage) runs about $4 for a 30-day supply. The brand? Around $350. Amlodipine (generic for Norvasc)? $2 versus $400. These aren’t outliers - they’re the norm. The Congressional Budget Office estimates that generics save the average patient $1,500 to $2,000 per year. Over the last decade, they’ve saved the U.S. healthcare system more than $2.2 trillion.What About the Differences in Color, Shape, or Taste?
Yes, generics look different. They might be blue instead of yellow, oval instead of round, or have a different imprint. That’s because trademark laws prevent generics from copying the exact appearance of brand-name drugs. These differences are purely cosmetic. They don’t affect how the drug works. The same goes for inactive ingredients - things like fillers, binders, dyes, and preservatives. These make up about 90% of the pill’s weight but serve no therapeutic purpose. They help the pill hold its shape, dissolve properly, or stay stable on the shelf. In rare cases, someone might react to a specific inactive ingredient. For example, a person allergic to a certain dye might get a rash from one generic version but not another. But this is extremely uncommon - the FDA reports fewer than 0.05% of switches trigger such reactions. Pharmacists can help you find a version without that ingredient if needed.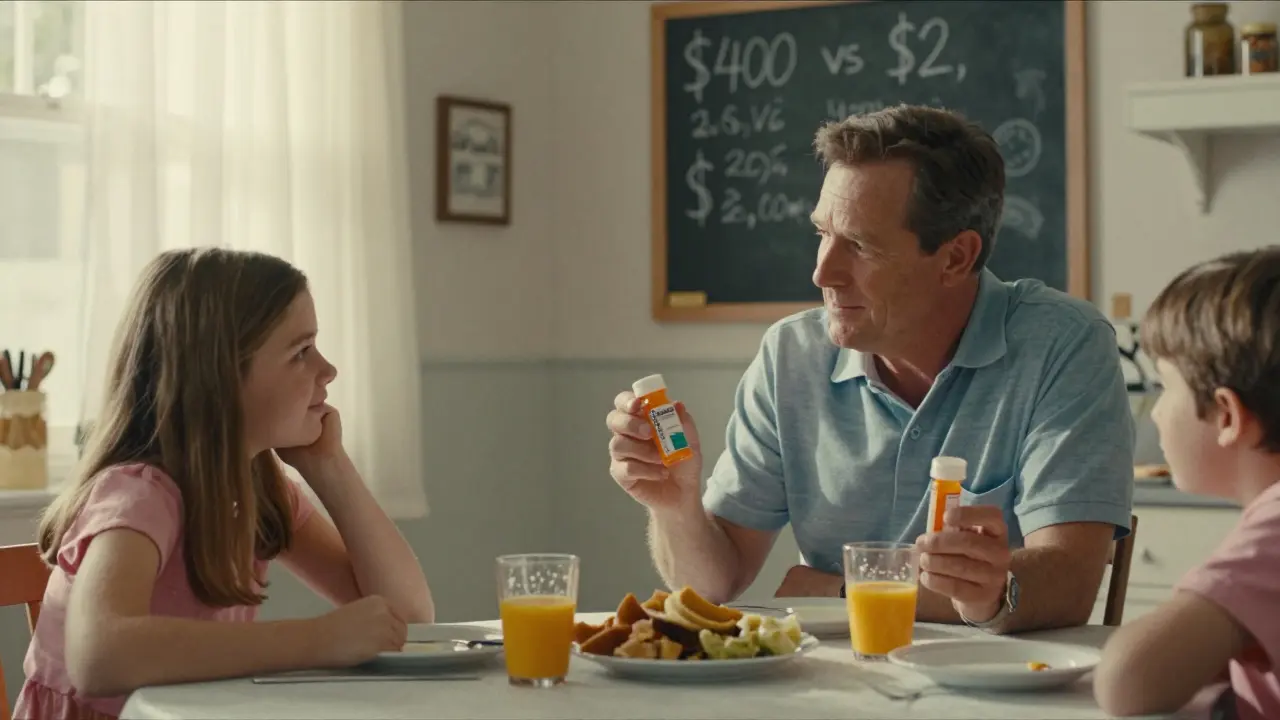
Are There Any Exceptions?
There are a few drugs where tiny changes in blood levels can matter. These are called narrow therapeutic index (NTI) drugs. Examples include warfarin (a blood thinner), levothyroxine (for thyroid disease), and some anti-seizure medications like phenytoin. For these, doctors sometimes prefer to stick with one version - brand or generic - to avoid even minor fluctuations. But even here, the FDA confirms that approved generics meet the same standards. If you’re on one of these medications and your doctor switches you to a generic, they’ll likely monitor you closely at first. That’s not because the generic is less effective - it’s just a precaution. A 2022 review in The New England Journal of Medicine analyzed 47 studies on cardiovascular generics. The conclusion? No meaningful difference in outcomes. Even for NTI drugs, the evidence shows generics are safe and effective when used as directed.What Do Patients Really Experience?
Real people report real results. On Drugs.com, generic medications have an average rating of 4.2 out of 5 stars from over 12,500 reviews. Most say they noticed no difference. One Reddit user wrote: “Switched from brand-name amlodipine to generic. Saved $300 a month. Blood pressure is the same.” But some report side effects. A few say they felt stomach upset or dizziness after switching. Often, these are temporary as the body adjusts. In other cases, it’s likely due to a change in inactive ingredients - not the active one. A 2023 GoodRx survey of 5,000 people found 87% saw no difference in effectiveness. Only 12% noticed mild changes, mostly gastrointestinal. And here’s the key: those who switched to generics were 28% more likely to keep taking their medication because they could afford it. Better adherence means better health outcomes - especially for chronic conditions like hypertension and diabetes.Can Pharmacists Substitute Generics Automatically?
In 49 out of 50 U.S. states, pharmacists can switch a brand-name prescription to a generic unless the doctor specifically says “do not substitute.” That’s because the law recognizes generics as therapeutically equivalent. Insurance companies encourage this too. Humana reports that 92% of brand-name requests are automatically filled with generics unless medically justified. Many plans won’t cover the brand unless you’ve tried and failed with the generic first. If you’re unsure about a switch, ask your pharmacist. They have access to the FDA’s therapeutic equivalence database and can explain why the generic is a safe replacement.
